|
The researchers tested their system using multiple datasets and found that it outperformed other AI systems that had tried to do the same-it achieved a 96 percent correlation for short time spans and 64 percent for relaxation demonstrations (which in real-time would be thousands of years). If cooled slowly enough, it may instead transform into an ordered crystal. 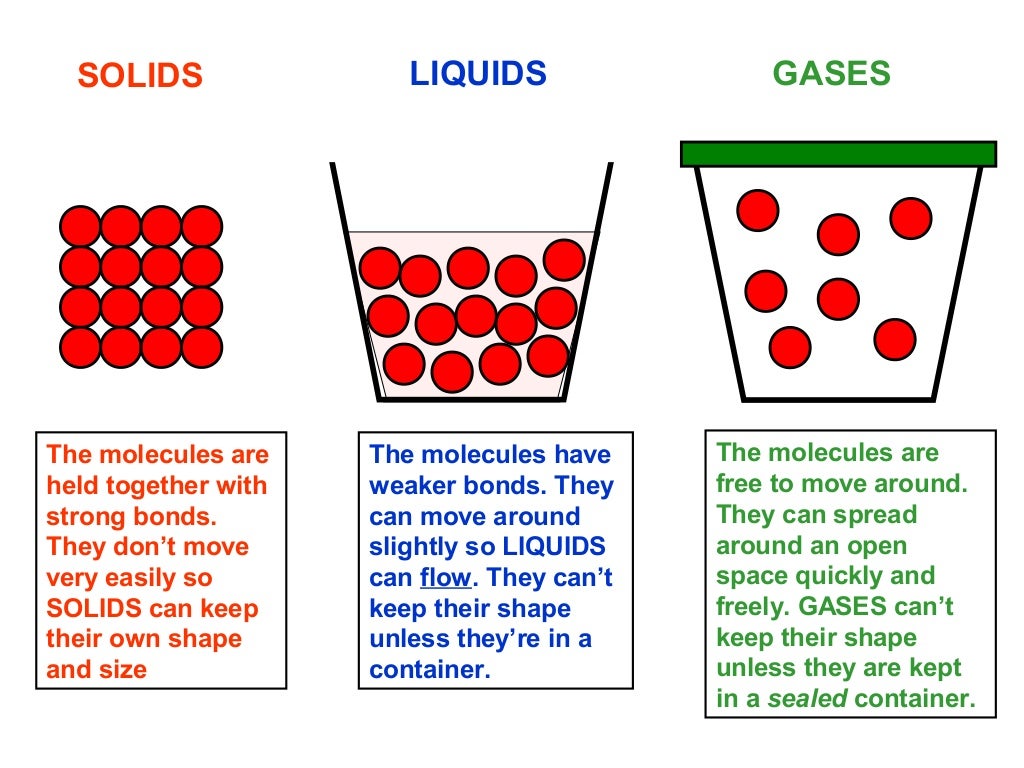
A liquid, when cooled too quickly past its crystallization point, turns into a supercooled liquid which, upon further cooling, turns into a disordered, amorphous glass. Once the AI system received the data, it was processed in a way that produced predictions of particle movement. The team also had to use an encoder to translate the particles and interactions to mathematical objects that could be recognized by the AI system. In such a system, particles were represented as connecting to nearby particles. To use such a system with glass, the team had to convert particles and interactions between them to nodes and edges. To gain some insight into the glass transition process, the researchers used graph neural networking, in which AI is used to develop systems that can work with graphs-nodes and edges-to make predictions about dynamic systems. Understanding the process would have an impact on more than just the glass industry it would also explain other similar transition processes, such as colloidal suspensions, granular materials and even cell migration. Intriguingly, despite many years of study, scientists do not really understand the nature of glass or its transition process. Also, when glass is made, something else happens that is of great interest to scientists-its viscosity increases a trillion-fold as it cools to a solid. Notably, the molecular structure of glass has no structure at all-under a microscope, the molecules appear to be assembled randomly. The end result of the process is a material that is hard, brittle and allows light to pass through easily. 
Sand and other silica components are heated to a very high temperature at which they melt, and the resulting material is then rapidly cooled past its crystallization point. Over those many years, improvements to the process have resulted in the development of many types of glass-but the basic process remains the same. Humans have been making glass for approximately 4,000 years. They have published a paper outlining their work on the DeepMind website. Credit: DeepMindĪ team of researchers at Google's DeepMind has developed an AI system that is able to predict the movement of glass molecules as the material transitions between liquid and solid states. The dynamics are heterogeneous – particle mobilities are correlated locally, but heterogeneous at macroscopic scales – yet our network still makes predictions in agreement with the ground truth simulation. In the right panel, corresponding to a timescale 28,000 times longer than the left panel, particles in the glass have started to diffuse. The left panel corresponds to a prediction over a short timescale: a regime in which our network attains a very strong performance. Better performance corresponds to greater alignment of red areas and dots. We call these molecules.GNN-predicted mobilities (colored from least mobile in blue to most mobile in red) compared to the position of the most mobile particles in the simulation (dots) in a slice of our 3-dimensional box. More usually they are groups of atoms bonded together, each group of atoms being the same. 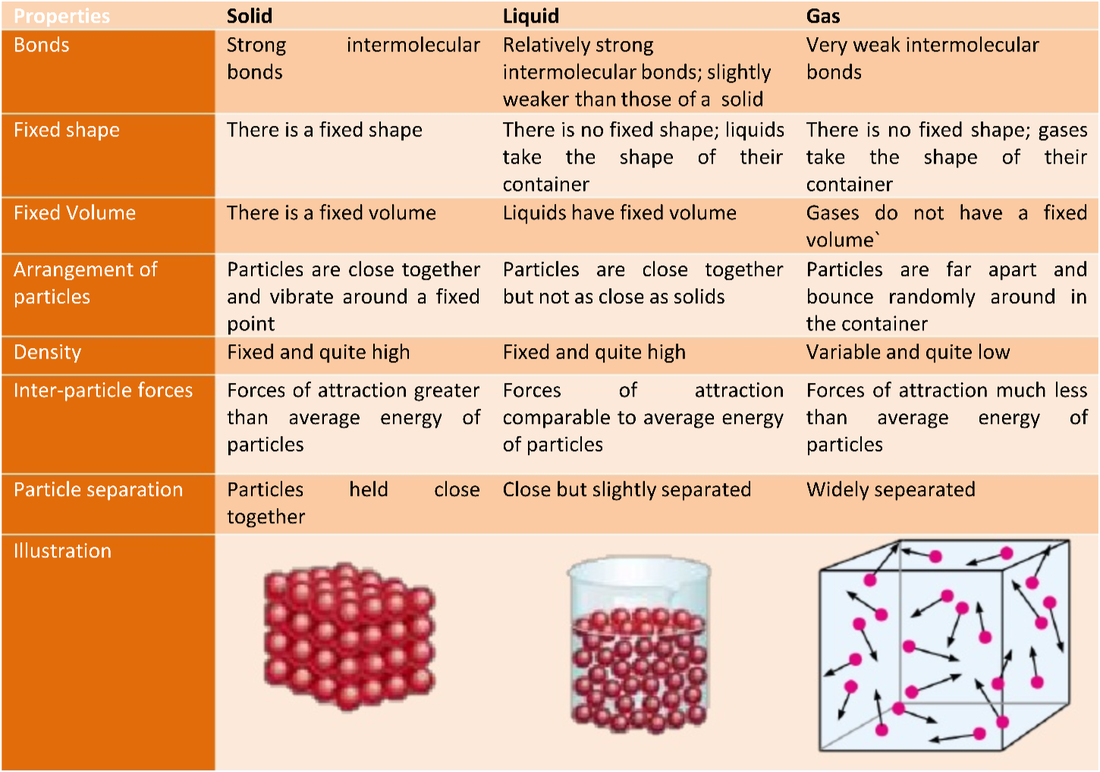
In some substances, they could be individual atoms e.g. 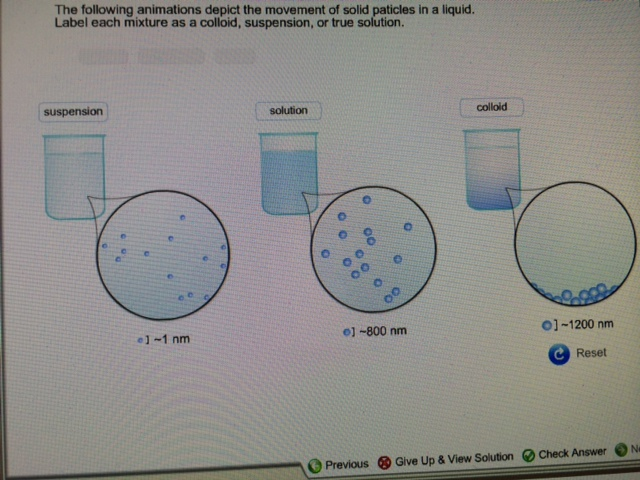
Because the particles are so far apart, they can be squeezed into a smaller volume, i.e. The particles can go anywhere within their container, and so gases have no shape. They have the most energy of the three states. Particles in the gaseous state are widely spread out and can both vibrate and move around freely. Because the particles in a liquid are still close together, liquids cannot be compressed. For this reason the particles in a liquid can flow, and a liquid will take up the shape of whatever container it is placed in. Particles in the liquid state are still closely packed, but can both vibrate and move around within the liquid because they have more energy – enough to overcome the forces that hold the particles together in the solid. Because the particles are so close together, solids cannot be compressed (squeezed into a smaller volume). Solids therefore have their own shapes because the particles do not flow. Particles in the solid state are closely packed and can vibratebut cannot move around, they have low energies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
